All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional.
The PsOPsA Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the PsOPsA Hub cannot guarantee the accuracy of translated content. The PsOPsA Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The PsOPsA Hub is an independent medical education platform. This activity is supported by an educational grant from Lilly. The funders are allowed no direct influence on our content.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View psoriasis and psoriatic arthritis content recommended for you
Efficacy and safety of bimekizumab compared with other available treatments in PsA: A network meta-analysis
Bimekizumab is an interleukin 17 (IL-17) inhibitor and is approved in Europe for use in psoriatic arthritis (PsA) in adults.1 Bimekizumab has shown efficacy in the phase III trials BE OPTIMAL (disease-modifying anti-rheumatic drug [DMARD]-naïve patients) and BE COMPLETE (tumor necrosis factor inhibitor [TNFi] inadequate responder patients), which has previously been covered on the Psoriasis and Psoriatic Arthritis Hub.1
Here, we summarize a network meta-analysis by Mease et al.1 in Oxford Rheumatology, which evaluated the efficacy and safety of bimekizumab for patients with PsA compared with 21 other biologic or targeted synthetic DMARDs (b/tsDMARDs).1
Methods1
- The meta-analysis (latest update on January 1, 2023) was designed to identify studies evaluating the efficacy and safety of bimekizumab 160 mg every 4 weeks (Q4W) vs other available PsA treatments.
- Two analyses were performed: one for b/tsDMARD-naïve and one for TNFi-experienced patients with PsA.
- In each patient population, the American College of Rheumatology 20%/50%/70% improvement responses (ACR20/50/70), Psoriasis Area and Severity Index (PASI) score of 90/100, and minimal disease activity score were analyzed. Serious adverse events analysis was conducted in a mixed population.
- Studies were selected if data were available at Week 16; if not, data at Weeks 12, 14, or 24 were used.
- Surface under the cumulative ranking curve (SUCRA) values were used to determine relative rank of treatments.
Key findings1
- Of the 66 studies selected for data extraction, 41 studies met the inclusion criteria.
Joint outcomes
- In b/tsDMARD-naïve patients, bimekizumab 160 mg Q4W was better in ACR50 (SUCRA = 0.74) than placebo, abatacept 125 mg, guselkumab 100 mg Q4W, ustekinumab 45 mg, risankizumab 150 mg, guselkumab 100 mg every 8 weeks, and ustekinumab 90 mg, but it was worse than golimumab 2 mg. Bimekizumab 160 mg Q4W ranked 6th for ACR20 (SUCRA = 0.75), 5th for ACR50 (SUCRA = 0.74), and 3rd for ACR70 (SUCRA = 0.80) among 21 treatments. Bimekizumab was comparable to the remaining treatments in the network.
- In TNFi-experienced patients, bimekizumab was ranked 1st among 16 treatments for ACR70 (SUCRA = 0.83) and was better than placebo, abatacept 125 mg, secukinumab 150 mg without loading dose, tofacitinib 5 mg, and secukinumab 150 mg; and it was comparable to the remaining treatments on ACR50.
Skin outcomes
- In b/tsDMARD-naïve patients, bimekizumab was ranked 1st of 11 treatments for PASI 100 (SUCRA = 0.95) (Figure 1A). In TNFi-experienced patients, bimekizumab was ranked 2nd of 7 treatments for PASI 100 (SUCRA = 0.79) (Figure 1B).
Minimal disease activity score
- Bimekizumab was ranked the 1st among the 13 treatments in b/tsDMARD-naïve patients (SUCRA = 0.91) and 11 treatments in TNFi-experienced patients (SUCRA = 0.83).
Safety
- Safety outcomes were found to be comparable across all treatments evaluated
Figure 1. A. Bimekizumab 160 mg Q4W vs other treatments for PASI 100 at Week 16 in A b/tsDMARD-naïve and B. TNFi-experienced patients*
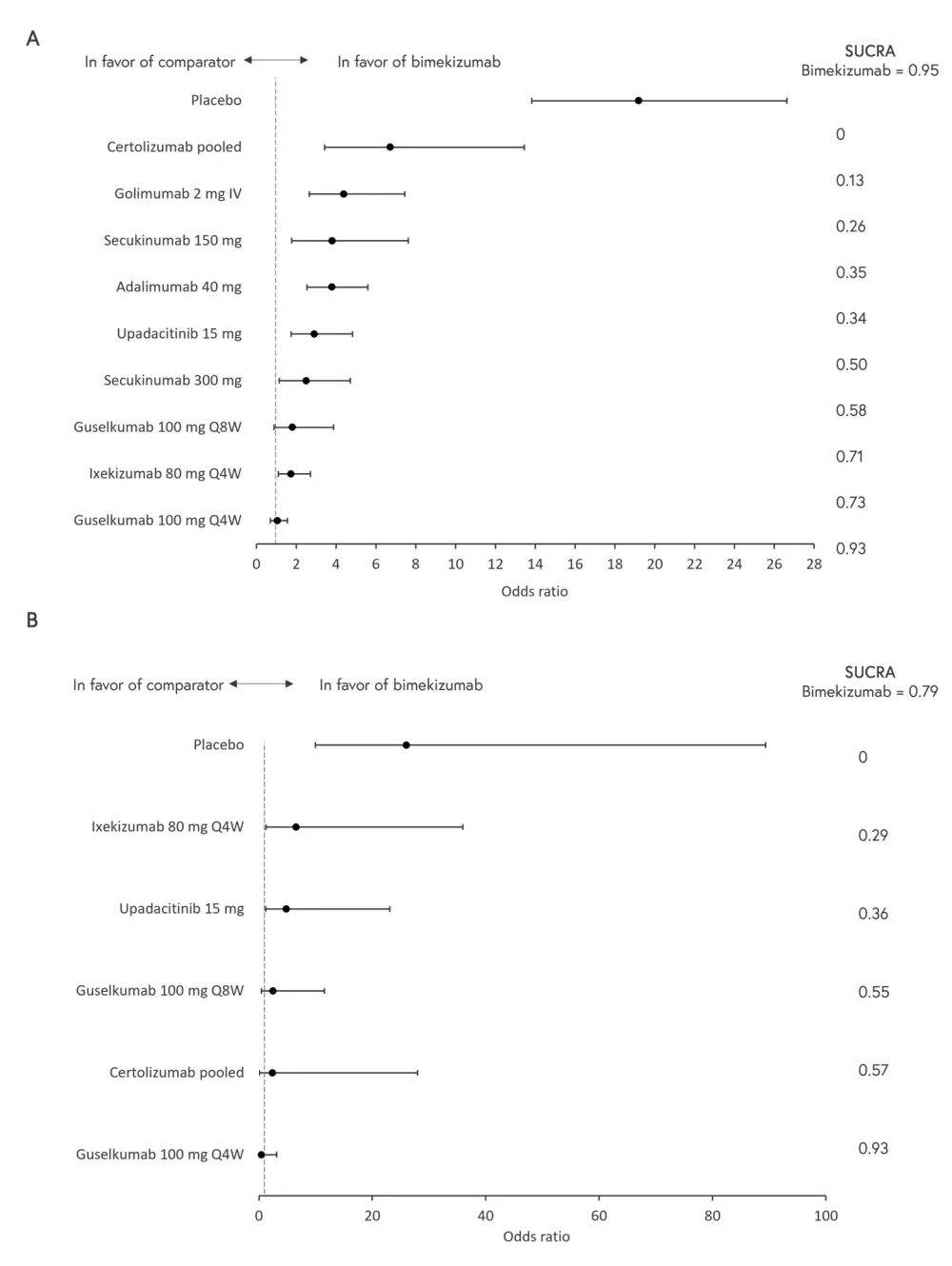
b/ts DMARD, biological or targeted synthetic disease-modifying anti-rheumatic drug; Q4W, every 4 weeks; Q8W, every 8 weeks; PASI, Psoriasis Area and Severity Index; SUCRA, surface under the cumulative ranking curve; TNFi, tumor necrosis factor inhibitor.
*Adapted from Mease, et al.1
|
Key learnings |
|
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content



