All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional.
The PsOPsA Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the PsOPsA Hub cannot guarantee the accuracy of translated content. The PsOPsA Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The PsOPsA Hub is an independent medical education platform. This activity is supported by an educational grant from Lilly. The funders are allowed no direct influence on our content.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View psoriasis and psoriatic arthritis content recommended for you
BE OPTIMAL and BE COMPLETE: 52-week post-hoc analysis of patients with BSA ≥3% and ≥10%
During the European Academy of Dermatology and Venereology Congress (EADV) 2023 Congress, Thaçi presented a post-hoc analysis of two phase III trials, BE OPTIMAL (NCT03895203) and BE COMPLETE (NCT03896581), of bimekizumab in patients with psoriatic arthritis (PsA). The analysis focused on the 52-week safety and efficacy of bimekizumab versus placebo in patients with PsA and psoriasis with ≥3% body surface area (BSA) affected at baseline.
The study design and baseline patient characteristics have been previously reported on the Psoriasis and Psoriatic Arthritis Hub.
Results
Safety
The safety outcomes for patients treated with bimekizumab 160 mg every 4 weeks are shown in Table 1. The most frequent treatment-emergent adverse events were nasopharyngitis and coronavirus infection in BE OPTIMAL and BE COMPLETE, respectively.
Table 1. Adverse events*
|
TEAE, treatment-emergent adverse event. |
||
|
TEAE, % |
BE OPTIMAL (N = 356) |
BE COMPLETE (N = 255) |
|---|---|---|
|
Any TEAE |
75.0 |
55.7 |
|
Severe TEAEs |
3.4 |
4.7 |
|
Study discontinuation due to TEAEs |
2.8 |
3.5 |
|
Drug-related TEAEs |
25.3 |
20 |
|
Serious TEAEs |
5.1 |
5.9 |
|
Deaths |
0.3 |
0.4 |
|
Serious infections |
0.6 |
2.0 |
Efficacy
Key efficacy outcomes, including 50% improvement in American College of Rheumatology response criteria, Swollen Joint Count ≤1, and Psoriasis Area and Severity Index ≤1, in patients with a BSA ≥3% and ≥10% are shown in Figure 1.
Figure 1. Patients achieving A ACR50, B SJC ≤1, and C PASI ≤1*
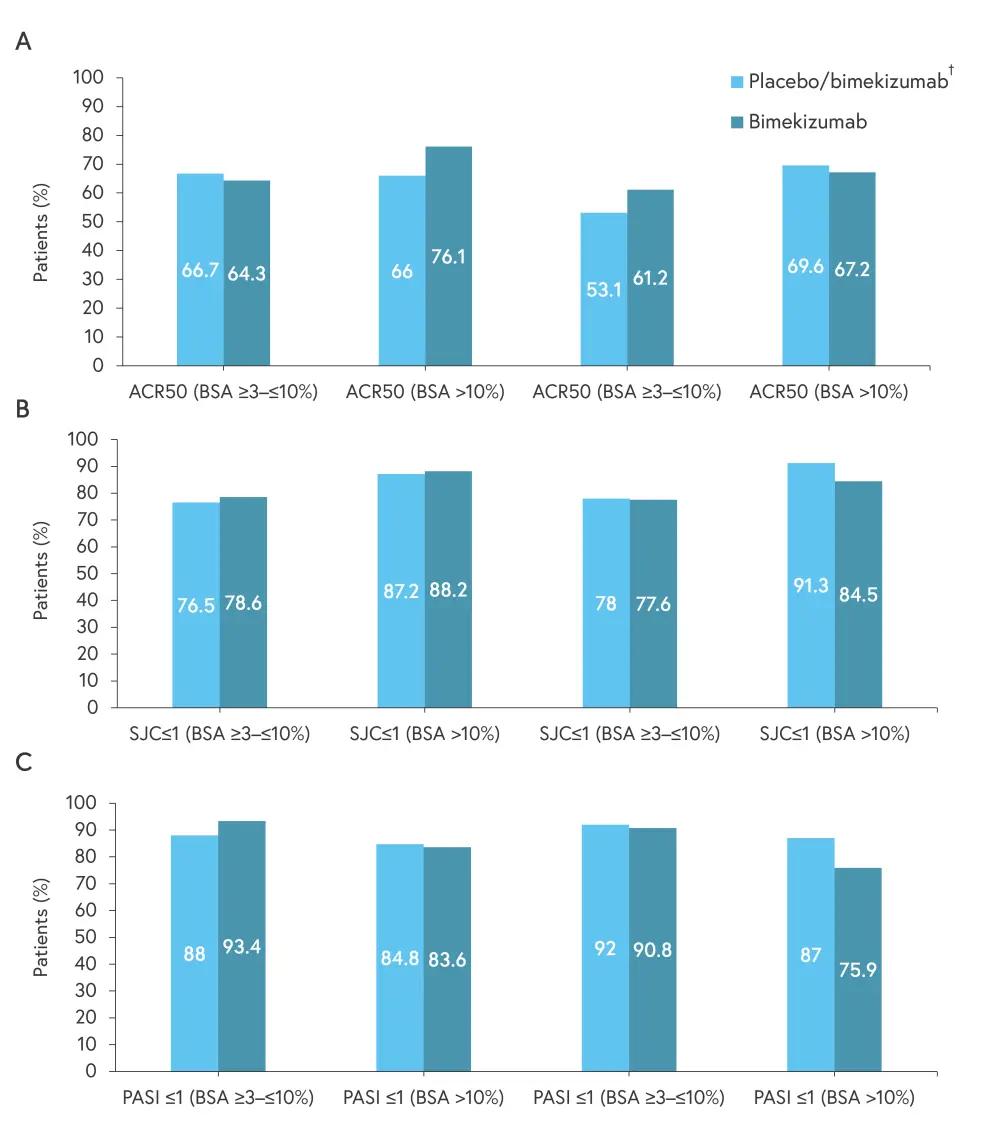
ACR50, 50% improvement in American College of Rheumatology response criteria; BSA, body surface area; PASI, Psoriasis Area and Severity Index; SJC, Swollen Joint Count.
*Data from Thaçi.1
†Patients randomized to placebo switched to bimekizumab at Week 16.
Conclusion
Week-52 results from BE OPTIMAL and BE COMPLETE demonstrated positive efficacy and safety outcomes with bimekizumab for patients with PsA and severe skin symptoms (BSA ≥3% and ≥10%). Similar response rates were seen across patients who were originally randomized to bimekizumab and those who switched from placebo to bimekizumab at Week 16.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

