All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional.
The PsOPsA Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the PsOPsA Hub cannot guarantee the accuracy of translated content. The PsOPsA Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The PsOPsA Hub is an independent medical education platform. This activity is supported by an educational grant from Lilly. The funders are allowed no direct influence on our content.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View psoriasis and psoriatic arthritis content recommended for you
Deucravacitinib vs apremilast and placebo for treatment of moderate-to-severe plaque psoriasis: The POETYK-PSO-2 trial
Given the safety and tolerability profiles of currently available biologics and small-molecule oral agents for plaque psoriasis, there is a need for novel, better-tolerated therapies. Deucravacitinib is an oral, selective tyrosine kinase 2 (TYK2) inhibitor that binds to the regulatory or pseudokinase domain of TYK2 and allosterically inhibits the enzyme from initiating cytokine synthesis and plaque psoriasis pathogenesis.
Here, we summarize the results of POETYK-PSO-2, a phase III, randomized, double-blind study assessing the efficacy and safety of deucravacitinib against apremilast and placebo for the treatment of moderate-to-severe plaque psoriasis (NCT03611751),1 published in the Journal of the American Academy of Dermatology by Strober et al.
Methods1
- POETYK PSO-2 trial was a 52-week, double-blinded, phase III study conducted across 191 sites in 16 countries between June 2018 and November 2019, and included 1,020 adult patients with moderate-to-severe plaque psoriasis and:
-
- Psoriasis Area and Severity Index (PASI) ≥12;
- static Physician’s Global Assessment (sPGA) ≥3; and
- body surface area involvement ≥10% for ≥6 months.
- Patients were randomized to deucravacitinib 6 mg every day (n = 511), placebo (n = 255), or apremilast 30 mg twice a day (n = 254).
- The coprimary efficacy endpoints at Week 16, for patients treated with deucravacitinib vs placebo, were:
-
- ≥75% reduction from baseline in PASI (PASI 75); and
- sPGA score of 0 (clear) or 1 (almost clear), with a ≥2-point improvement from baseline (sPGA 0/1).
- Secondary endpoints included Week 16 PASI 90, PASI 100, sPGA 0 vs placebo, and Week 24 PASI 90 vs apremilast.
Key findings1
- More patients who received deucravacitinib achieved the coprimary endpoints of PASI 75 and sPGA 0/1 at Week 16 than patients who received apremilast or placebo (Figure 1).
- Deucravacitinib was also associated with higher PASI 90, PASI 100, and sPGA 0 responses at Week 16 and a higher PASI 90 vs apremilast at Week 24.
- Rates of adverse events (AEs) and serious AEs were similar in patients who received deucravacitinib and those who received apremilast; the most common AEs were nasopharyngitis and upper respiratory tract infections in the deucravacitinib group, and headache, diarrhea and nausea in the apremilast group.
Figure 1. Efficacy measures at the end of Week 16*
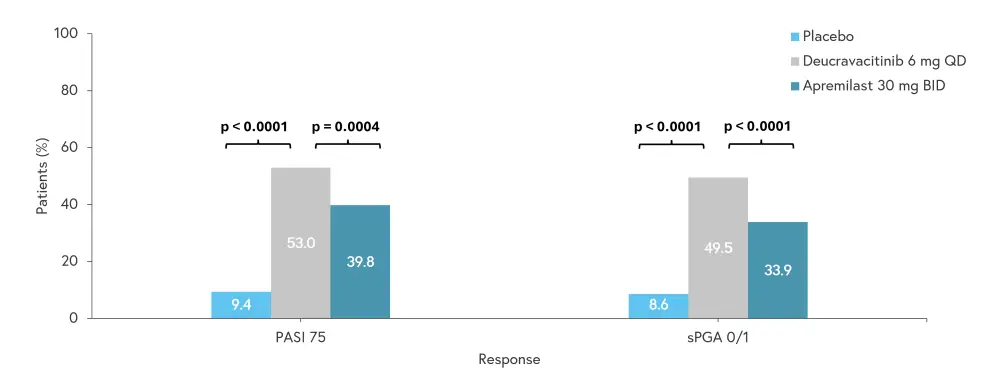
BID, twice daily; PASI, Psoriasis Area and Severity Index; QD, once daily; sPGA, static Physician’s Global Assessment.
*Data from Strober, et al.
| Key learnings |
|
Deucravacitinib demonstrated clinically significant efficacy over placebo and apremilast in adults with moderate-to-severe plaque psoriasis over 52 weeks. |
|
Deucravacitinib was generally well tolerated, with a safety profile consistent with the mechanism of selective TYK2 inhibition, showing low rates of serious AEs. |
|
This trial suggests that deucravacitinib may be a viable oral, once-daily treatment option for patients with plaque psoriasis, although longer-term data are needed. |
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content


