All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional.
The PsOPsA Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the PsOPsA Hub cannot guarantee the accuracy of translated content. The PsOPsA Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The PsOPsA Hub is an independent medical education platform, supported by educational grants. We would like to express our gratitude to the following companies for their support: UCB, founding supporter. The funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View psoriasis and psoriatic arthritis content recommended for you
Biologics in pregnant women with psoriasis: Real-life practice of dermatologists
The management of plaque psoriasis during pregnancy is not well understood. The Summaries of Product Characteristics (SmPC) for biologics recommend discontinuation during pregnancy, and most should be stopped up to 24 weeks prior to conception. Only certolizumab pegol may be used in pregnancy if appropriate. In France, official guidelines follow the recommendations of the SmPCs; however, practitioners may not always follow these recommendations.
Here, we summarize a study by Tran et al.1 published in Annales of Dermatology and Venereology on the real-life practice of dermatologists in the management of pregnant women with psoriasis treated with biologics.
Study design1
- A retrospective, observational, multicenter study by two dermatologist research groups in France.
- Practitioners were sent a two-part survey for completion between March and June 2021. The first part included 6 questions about their profile and the second part included 12 questions assessing their clinical practice concerning the use of biologics during pregnancy.
- Practitioners were also assessed for their confidence with continuing different biologics during pregnancy, using a 10-point Likert scale.
- The following biologics were studied in the survey:
-
- Etanercept
- Infliximab
- Adalimumab
- Certolizumab pegol
- Ustekinumab
- Brodalumab
- Ixekizumab
- Secukinumab
- Risankizumab
- Guselkumab
Key findings1
- A total of 72 practitioners took part in the study, with a mean age of 43.8 years. The majority were female (71.4%) and hospital-based.
- Less than half of practitioners surveyed discontinued biologics before conception as per the SmPC guidelines, with the majority of practitioners discontinuing biologics when pregnancy was confirmed (Figure 1).
- For practitioners with more than 10 years of experience in treating psoriasis, 81% reported not following the SmPC recommendations (p = 0.023).
- Practitioners were most confident in continuing anti-tumor necrosis factor antibodies during pregnancy, with the highest mean confidence index level with certolizumab (9/10), followed by etanercept (7/10) and adalimumab (7/10).
-
- Practitioners were least confident in continuing anti-interleukin 17 and 23 antibodies (mean level of confidence = 3/10).
Figure 1. Management strategies of practitioners for biologic use in psoriasis during pregnancy*
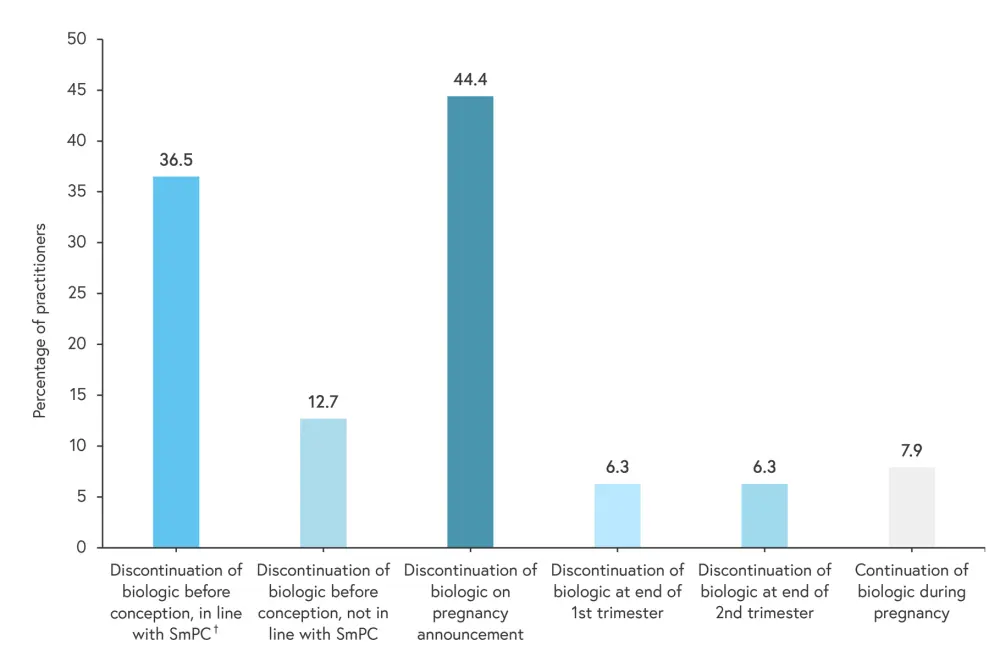
SmPC, Summaries of Product Characteristics.
*Adapted from Tran, et al.1
†The SmPC for biologics recommend discontinuation during pregnancy and should be stopped up to 24 weeks prior to conception.
|
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

