All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional.
The PsOPsA Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the PsOPsA Hub cannot guarantee the accuracy of translated content. The PsOPsA Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The PsOPsA Hub is an independent medical education platform. This activity is supported by an educational grant from Lilly. The funders are allowed no direct influence on our content.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View psoriasis and psoriatic arthritis content recommended for you
Guselkumab for scalp psoriasis: Analysis of VOYAGE-2
Approximately 80% of individuals with psoriasis experience scalp involvement, which is associated with increased disease severity, and decreased quality of life.1 Despite the available treatment options including topicals, phototherapy, systemic therapy, biologics, and oral small molecules, scalp psoriasis remains difficult-to-treat.1 VOYAGE-2 was a phase III, multicenter, randomized trial, which evaluated guselkumab, anti-IL-23 antibody, compared with placebo and an active comparator (adalimumab).1
Here, we summarize a post hoc analysis of VOYAGE-2 by Sonkoly et al.1 published in Acta Dermato-Venereologica, that evaluated scalp response and its association with skin response and patient-reported outcomes.
Study design1
- This analysis included three subsets of patients with scalp psoriasis:
-
-
- Responder continuation group, who achieved a 90% improvement in the Psoriasis Area and Severity Index (PASI90) response at Week 28 and continued guselkumab
- Non-responder continuation group, who did not achieve a PASI90 response at Week 28 but continued guselkumab
- Responder withdrawal group, who achieved a PASI90 response at Week 28 and were re-randomized to placebo
-
- Skin efficacy was assessed by total PASI scores and scalp efficacy using the scalp-specific Investigator’s Global Assessment (ss-IGA).
- Patient-reported outcomes were measured using the Psoriasis Symptoms and Signs Diary (PSSD) and Dermatology Life Quality Index (DLQI).
Key findings1
- A total of 407 patients with moderate-to-severe plaque psoriasis were included.
-
- The majority of patients were male (71.8%).
- Mean ss-IGA of 2.9, the mean PASI scores and mean DLQI were similar across the subsets.
- Total PASI scores and head PASI scores improved from baseline to Week 24 across all subgroups.
- Mean ss-IGA scores improved from baseline to Week 24, and remained stable to Week 48 in all subgroups, while the scores worsened in the responder withdrawal group from Week 24 through Week 48 (Figure 1).
- Similarly, total PASI and head PASI scores remained stable from Week 24 to Week 48 across all subgroups apart from the responder withdrawal group.
- Mean PASI head score remained stable from Week 48 (0.1) to Week 252 (0.1) in the responder continuation group and improved in the non-responder continuation group (0.3 and 0.1 at Weeks 48 and 252, respectively).
- In the responder withdrawal group, the mean PASI head score worsened after treatment withdrawal at Week 28 (0.1) through Week 48 (0.6) and improved slightly through Week 72 (0.5). After retreatment with guselkumab at Week 72, scores improved rapidly through Week 84 (0.1) and remained stable through Week 252 (0.1).
- Quality of life, as measured by DLQI, and PSSD itch scores, improved across all subgroups from baseline to Week 24.
- Within-subject correlations were 0.83 between ss-IGA and PASI head scores and 0.78 between ss-IGA and PSSD itch scores.
Figure 1. Changes in mean ss-IGA scores from baseline to Week 48 in the three scalp subgroups*
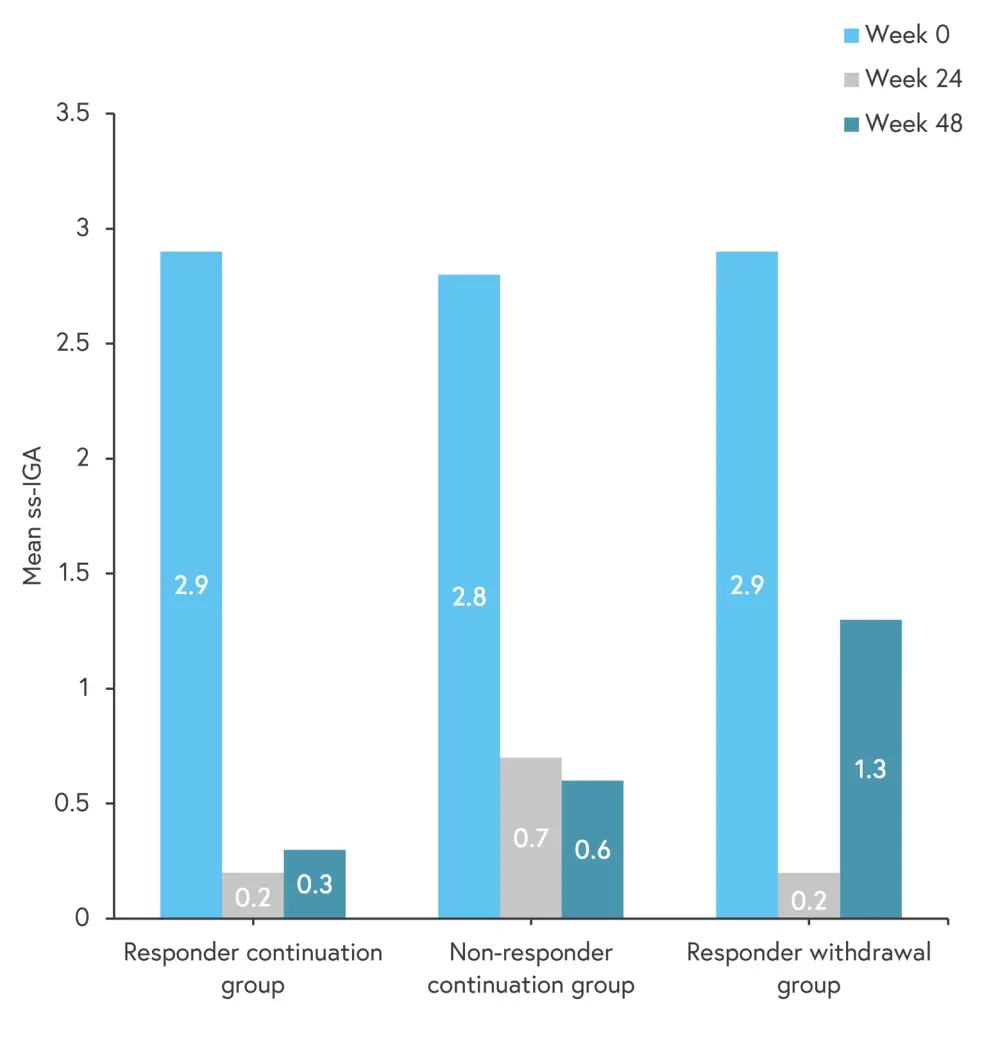
ss-IGA, scalp-specific Investigator’s Global Assessment.
*Data from Sonkoly, et al.1
| Key Learnings |
|
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

